10 Ways We Helped Our Parkinson’s Community This Year
Together, we made life better for people in our Parkinson’s community in 2019. Your support allowed us to launch new, exciting initiatives that are changing lives, while funding critical research and local classes tailored to people with Parkinson’s disease (PD). With your support, we are reaching more people living with this disease in the U.S. and are closing the gap to help the 60,000 Americans diagnosed every year.
Thanks to YOU, we were able to accomplish the following in 2019:
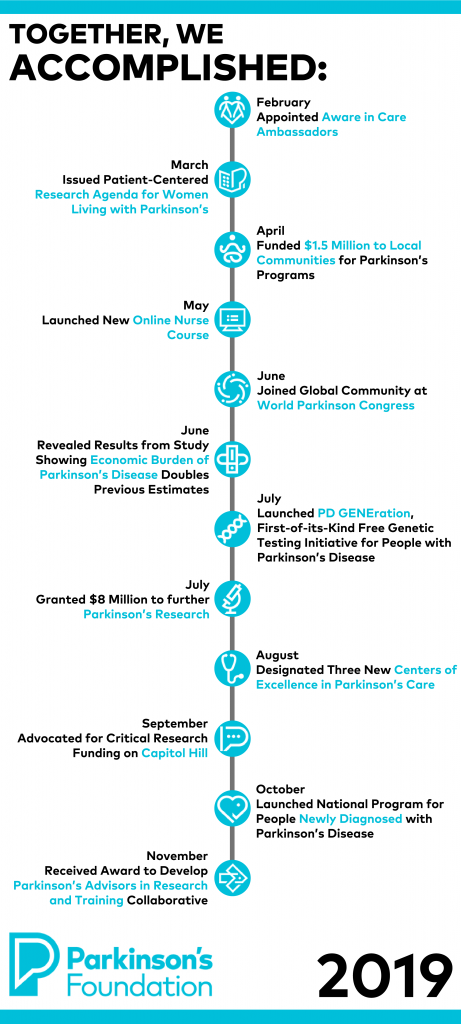
- Launched First-of-its-Kind Free Genetic Testing Initiative
We launched PD GENEration: Mapping the Future of Parkinson’s Disease, a national initiative that offers free genetic testing for clinically relevant PD-related genes and free genetic counseling. We are excited to reach our goal of testing and providing genetic counseling for up to 15,000 people with PD.
- Funded $12.2 Million to Further Parkinson’s Research
This was an impactful year for Parkinson’s research. We established four new Research Centers that will receive a total of $8 million to launch PD-specific research studies. We also simultaneously funded $4.2 million across 46 research grants that support the work of promising scientists in the PD field, cutting-edge clinical trials and fellowships.
- Designed Program for People Newly Diagnosed with Parkinson’s
With a focus to reach the 60,000 people who are newly diagnosed with PD each year in the U.S., Newly Diagnosed: Building a Better Life with Parkinson's Disease aims to close the gap between diagnosis and knowing where and how to find the right information and resources to live better with PD. Order or download the free Newly Diagnosed kit. - Shared Research Findings at the 5th World Parkinson Congress
In June, we enthusiastically joined our international community at the 5th World Parkinson Congress (WPC) in Kyoto, Japan, where our PD experts shared 10 research posters.
- Expanded our Centers of Excellence Care Network
Every year, we hope to bring expert care to more people with PD. In 2019, we expanded our Center of Excellence global network to include three new centers. Centers of Excellence are a medical center with a specialized team who provide expert Parkinson’s care. Find a Center of Excellence in your area.
- Partnered with Michael J. Fox Foundation for Parkinson’s Research to Report New Economic Burden and Host Policy Forum
In 2019, we collaborated with the Michael J. Fox Foundation (MJFF) to publish that the economic burden of Parkinson’s is nearly $52 billion every year. Together, we also hosted the 2019 Parkinson’s Policy Forum, bringing more than 150 advocates from across the U.S., along with leading experts in PD research to advocate for our community.
-

Granted $1.5 Million to Local Communities for Parkinson’s Programs
We proudly funded $1.5 million throughout 118 community-based grants that provide education and outreach programs, along with local research initiatives, that address unmet needs in the PD community. - Issued First Patient-Centered Research Agenda for Women with PD
Recognizing long-standing gender disparities in Parkinson’s research and care, our Women and Parkinson’s Initiative created the first patient-centered action agenda to maximize quality of life for women with Parkinson’s.
- Appointed Aware in Care Ambassadors
In 2019, we appointed our first-ever Aware in Care Ambassadors, a volunteer group to help distribute Aware in Care kits that serve to bolster best practices in treating patients with Parkinson’s disease to both patients and healthcare providers.
- Designed New Online Nurse Course
With the prevalence of PD expected to increase in the coming years, we wanted to provide more professional education opportunities for nurses. In 2019, we launched a new online course for nurses who deliver care throughout all stages of Parkinson’s.
As much as we accomplished in 2019, we are committed to furthering our reach and impact in 2020 to help even more people live better with Parkinson’s. Your continued support is the only way we can make that happen. Thank you.
Related Blog Posts

Caring for the Mind: 10 Parkinson’s Mental Wellness Resources

People with Parkinson’s Share What’s Helped Them