Clinical Trials
💡 Quick Summary
-
Clinical trials are research studies that test potential new treatments, therapies or approaches in people.
-
Clinical trials do not replace standard Parkinson’s care. Most people continue their usual treatment while participating.
-
Clinical trials can lead to better treatments and care for people with Parkinson’s.

Clinical trials are research studies that test new treatments and approaches to care in people. In Parkinson’s disease (PD), clinical trials help researchers learn what is safe and what works, and are an important step in developing new treatments. Each study is different, and participation is always voluntary.
Current Treatments vs. Clinical Trials
Clinical trials are different from the treatments your care team recommends, but they are closely connected. The treatments your care team recommends, also called the standard of care, are widely used by doctors because research and experience show they help manage symptoms. These treatments are part of everyday care for people with Parkinson’s.
An intervention is the treatment or approach being studied in a clinical trial. Interventions may include medications, devices, therapies or structured programs such as exercise or speech therapy.
Some trials focus on controlling symptoms, while others explore ways to slow or stop the disease. Taking part in a clinical trial does not replace the treatments your care team recommends. Most people continue their usual care while participating.
Clinical trials are an important step in finding out if new treatments are safe and effective before they become part of standard Parkinson’s care.
Current Treatments ("Standard of Care") |
Clinical Trials |
|---|---|
| Manage symptoms with treatments known to work | Test new treatments or new uses for existing treatments |
| Supported by research and by medical experience | Being studied to see if they are safe and effective |
| Prescribed and available through your care team | Only available to study participants |
| Adjusted to fit your needs | Participants follow strict study rules and procedures |
| Ongoing part of your care | Usually done alongside your regular care |
How are new treatments approved?
The U.S. Food and Drug Administration (FDA) requires new treatments to go through three phases of clinical trials. After approval, some treatments enter a fourth phase. Each phase focuses on a key question about the treatment.
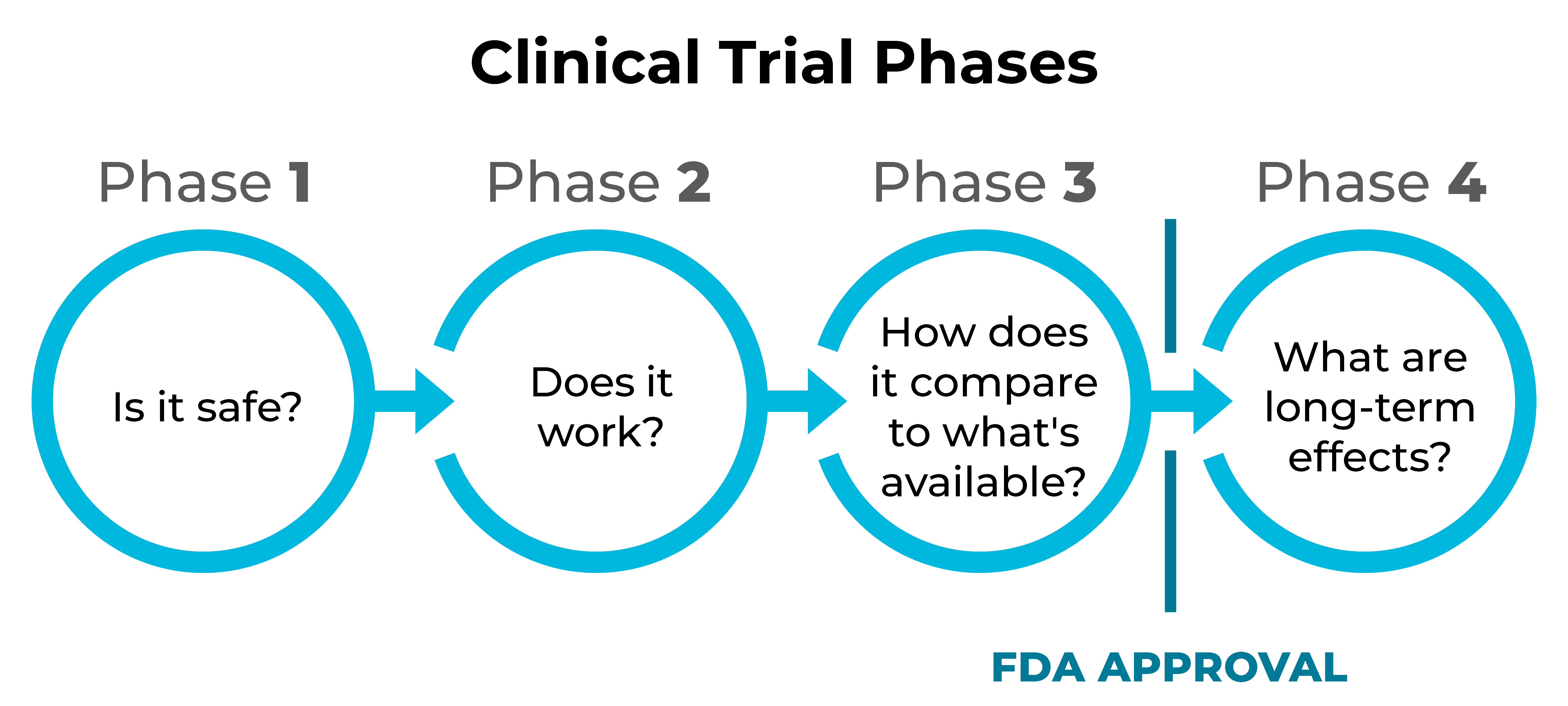
Is a clinical trial right for me?
Deciding whether to explore a clinical trial is a personal choice. It depends on your goals, your health and how comfortable you feel with research.
Some people join clinical trials to try a new treatment that might help them. They may also want to learn more about their Parkinson’s or contribute to research that may benefit others. Others may choose to focus on their current treatment plan instead.
Take time to ask questions and talk with your care team. This can help you decide if a clinical trial feels right for you. Then consider these factors as you make your decision and plan your next steps:
Types of Clinical Trials
What to Expect When You Participate in a Clinical Trial
Clinical trials follow a structured plan. While each study is different, many involve the following elements:
What are potential benefits and risks of participating in a clinical trial?
Clinical trials test new treatments, which means there can be both benefits and risks. Trials are carefully designed to protect participant safety, but uncertainty is part of research.
Possible benefits:
-
More contact with healthcare professionals, along with access to potential new treatments
-
Greater understanding of Parkinson’s and your own health
-
Contribution to research that may benefit others
Many current Parkinson’s treatments exist because people volunteered for clinical trials in the past.
Possible risks:
-
Side effects from the study treatment, including some that may not be known or expected
-
Lack of benefit from the treatment
-
A study schedule that may be difficult to fit into daily life

I have been in about 15 clinical trials, to help find new treatments for this awful disease. It gives me a real shot in the arm when I show up and meet the front-line researchers who are racing to find a cure or at least a treatment to slow the progression.

I immediately went to clinicaltrials.gov to see what studies I should sign up for, thinking I could learn more about my new situation, and how to get a handle on it.
Talk to Your Doctor About Clinical Trials
Your care team — especially your neurologist — can help you think through whether a clinical trial may be a good option. This conversation can clarify how a study relates to your diagnosis, symptoms, and treatment goals and what participation would involve. Talking with your doctor or the study team before joining a study can support an informed decision.
Questions to Ask
You may want to ask about the purpose of the study, what participation would involve, possible side effects, time commitment and how the trial compares with current treatment options.
Coordinating Care During a Trial
If you decide to participate in a clinical trial, your care team can help coordinate communication between the study team and those involved in your ongoing care.
Finding and Exploring Trials
There are several ways to learn about clinical trials for Parkinson’s and explore studies that may be a good fit for you. These websites list clinical trials and study details:
-
Join A Study — Parkinson’s Foundation resource that shares information about Parkinson’s research studies, including Foundation-supported trials
-
ClinicalTrials.Gov — A public database that lists active and completed clinical studies in the United States and around the world
Reviewing study details can help you understand your options and decide what you may want to explore next.
Haven’t started Parkinson’s medication yet?
Some clinical trials — especially those testing new treatments — look for people who have not started Parkinson’s medication (sometimes called “treatment naïve”). This helps researchers understand how a treatment works without the effects of other medications.
Our Research
The Parkinson’s Foundation study PD GENEration: Powered by the Parkinson’s Foundation focuses on the genetic links to Parkinson’s and offers genetic testing and counseling at no cost. Participants can learn whether genetics may play a role in their Parkinson’s and may also have additional ways to learn about clinical trial options through the PD Trial Navigator.
Information to Have Ready
When exploring trials, it can be useful to have your diagnosis date, current symptoms, medications and treatment history as well as other medical conditions that you have.
What to Do After Finding a Clinical Trial
After identifying a potential clinical trial, review the study details, talk with your care team and contact the study team to ask questions and learn more about participation. Reaching out to a study team does not mean you are committing to take part. You can ask questions and take time to decide. Even if you choose to join, you can stop at any time.
Make Sure a Trial is Legitimate
Be careful with trials that make big promises, skip over risks or ask for payment. Clinical trials do not charge people to take part. Trials should have approval from an institutional review board (IRB). You can request to see the IRB submission for more details on the study. Except for some low risk research like surveys and focus groups, the researcher should ask you to sign an informed consent form and walk you through the potential risks and benefits of participating before you sign the form.
Clinical Trials Are a Personal Choice
There is no obligation to take part in research. Not taking part does not affect access to Parkinson’s care, treatment options or benefits. Everyone’s situation is different. Decisions should reflect your needs, priorities and comfort levels.
Page reviewed by Dr. Michael Schwarzchild, Movement Disorders Neurologist at Massachussets General Hospital, Dr. Ruth Schneider, Movement Disorders Neurologist at Strong Memorial Hospital at the University of Rochester , and Dr, Roy Alcalay, Movement Disorders Neurologist at Tel Aviv Sourasky Medical Center
Join the Fight Against Parkinson's
Support ground-breaking research, care and community outreach